The Big Picture Podcasts
 For students, instructors, and life-long learners, our Big Picture Podcast series distills the flavor of the college experience through two formats: 1) The “Back Story” where we explore topics such as “how to study effectively”. We interview special guests including educators, scientists, and engineers. We also take the listener on audio field trips such as to the grounds of a former plutonium facility and a farm based upon sustainable agriculture. 2) “Do the Review” where the listener sits down with the textbook authors as they discuss the main ideas and nuances of each chapter of your “conceptual” textbook. A super-powered office visit sure to help you on your exams.
For students, instructors, and life-long learners, our Big Picture Podcast series distills the flavor of the college experience through two formats: 1) The “Back Story” where we explore topics such as “how to study effectively”. We interview special guests including educators, scientists, and engineers. We also take the listener on audio field trips such as to the grounds of a former plutonium facility and a farm based upon sustainable agriculture. 2) “Do the Review” where the listener sits down with the textbook authors as they discuss the main ideas and nuances of each chapter of your “conceptual” textbook. A super-powered office visit sure to help you on your exams.
Put this all together and we’re talking about a fun and engaging way to learn as well as appreciate basic science concepts anytime, anywhere. Mostly, this podcast series is a thoughtful step back—a reflection on the meaning of one’s education, which we call the big picture. We are starting with Conceptual Chemistry, which will be followed by podcasts of our other conceptual titles. Stay tuned!
Chem 101: Big Picture Podcast, Chapter 01, How to Study Effectively
Chapter 01 (Episode 01): Introduction and How to Study Effectively
Show Notes
This first episode presents effective study strategies, such as delayed retrieval, interleaving, and step 1 / step 2 learning. This is followed by a review of the first chapter of the Conceptual Chemistry textbook including topics such as the scientific method, basic and applied research, and unit conversion. Advice on how to make good use of the traditional science textbook is also provided.
References:
Make It Stick: The Science of Successful Learning, Peter C. Brown, et. al. The Belknap Press, 2014. ISBN: 978-0674729018.
How To Study Effectively, John Suchocki, 2018.
Transcript:
PDF: Chem101BPP01
Chem 101: Big Picture Podcast, Chapter 02, The Anthropology of Learning
Chapter 02 (Episode 02): The Anthropology of Learning
Show Notes
Description: From the authors of the “conceptual” line of science textbooks, in this second episode we discuss the how cognitive “frames” color our worldview. Recognizing these frames can help us to remain open to new ideas. The yet to be recognized term “collective causation” is used as an example of hypocognition. This is followed by a review of Chapter 2 of Conceptual Chemistry relating to the particulate nature of matter.
References: Cognitive Neuroscience: The Biology of the Mind, Michael Gazzaniga, Richard B. Ivery, George R. Mangun, W. W. Norton & Company, New York, NY, 2019.
Culture and the Individual: Theory and Method of Cultural Consonance, William W. Dressler, Routledge, New York, NY, 2018.
Don’t Think of an Elephant: Know Your Values and Frame the Debate, George Lakoff, Chelsea Green Publishing, White River Junction, VT, 2014.
Frames
“Draw the picture, build the frame Write the words, remember the names Everything that happened, happened as such Feather weight emotions just to touch You could never see it since you are there When outside die is beyond compare Eagles eye, end of the sky People see what they see, still ask why Frames of thought, frames of mind Trapped inside, trace the lines Old world, enter outside Own your truth, no need to hide No eyewitness I can see Fresh for the times, be who I be Make a timeline then break free Own yourself, you can never own me.: Noah Haspray April 2019 soundcloud.com/white-kong
Transcript: PDF: Chem101BPP02
Chem 101: Big Picture Podcast, Chapter 03, Team-based Learning
Chapter 03 (Episode 03): Team-based Learning
Show Notes
Description: From the authors of the “conceptual” line of science textbooks, in this third episode we interview Brian Pritchard an expert in the area of team based activities and human performance. Attention is given to the value of team-based learning in the classroom and beyond. This is followed by a review of Conceptual Chemistry’s foundational Chapter 3, Elements of Chemistry. Topics include physical and chemical changes, the periodic table, elements and compounds, mixtures, and nanotechnology.
References:
Anne Frank: The Diary of a Young Girl, Anne Frank, Doubleday & Company, New York, NY, 1946.
The Fifth Discipline: The art & Practice of The Learning Organization, Peter M. Senge, Penguin Random House, New York, NY, 2006.
“We will never transform the prevailing system of management without transforming our prevailing system of education. They are the same system.” W. Edwards Deming
Transcript:
PDF: Chem101BPP03
Chem 101: The Big Picture Podcast, Chapter 04a, Academic Endurance
Chapter 04a (Episode 04): Academic Endurance
Show Notes
We are now publishing the “Back Story” and “Do the Review” segments of each chapter as separate podcasts. This means the episode number will start to diverge from the chapter number. For this fourth backstory episode (Episode 04) we interview Stephanie Blake, a science professor from Ozarks Technical College in Springfield, Missouri. The subject matter: Academic Endurance. Learn from Professor Blake how best to strengthen your academic endurance. It has everything to do with working within a community and enjoying the journey. Duration: 30 min 54 sec.
References:
Year of Yes, How to Dance It Out, Stand in the Sun, and Be Your Own Person, Shondra Rhimes, Simon and Schuster Publishing, New York, 2016.
Transcripts:
Chem 101: The Big Picture Podcast, Chapter 04b, Subatomic Particles
Chapter 04b (Episode 05): Subatomic Particles
Show Notes
Here in episode 5 we review the first half of chapter 4 of Conceptual Chemistry. We start by reviewing the conceptual model, which describes how a system behaves. We then explore some of the history behind the discovery of subatomic particles. This episode concludes with a review of the vocabulary associated with subatomic particles, including atomic number and atomic mass number. Duration: 30:55.
Transcripts (pdf):
Chem 101: The Big Picture Podcast, Chapter 04c, Atoms and Quantum Theory
Chapter 04c (Episode 06): Atoms and Quantum Theory
Show Notes
This episode features a review of the second half of Chapter 4 of Conceptual Chemistry. It begins with the nature of light followed by the idea that energy itself is quantized, which means it occurs in packets. We then explore the wave nature of electrons and how this explains an element’s spectral fingerprint as well as the organization of the periodic table. Duration: 42:25.
Transcripts (pdf):
Chem 101: The Big Picture Podcast, Chapter 05a, Visit to Rocky Flats
Chapter 05a (Episode 07): Visit to Rocky Flats
Show Notes
For this “Back Story” segment for our nuclear chapter, we visit the site of the former Rocky Flats plutonium facility where from 1952 to 1989 plutonium triggers were produced for the hydrogen bombs in the USA nuclear arsenal. Major anti-nuclear protests were held at this site in the 1980s leading, in part, to its closure. Through decades of operation, much plutonium and other toxic chemicals were released into the local area, which is now a dedicated wild life refuge. We provide some of the history of Rocky Flats and even dig into some of the science behind the building of nuclear bombs. However, given the magnitude of significance, we also aim to provide space for reflection as supported by the audio nuances captured by our field recorder. Duration: 27:15.
[Coordinates: 39.883312, -105.235203]
Online References: NuclearCarePartners.com
RockyFlatsNuclearGuardianship.org
https://www.fws.gov/refuge/rocky_flats/
Show Transcripts: Chem101BPP05a
Chem 101: The Big Picture Podcast, Chapter 05a, Visit to Rocky Flats
Chapter 05a (Episode 07): Visit to Rocky Flats
Show Notes
For this “Back Story” segment for our nuclear chapter, we visit the site of the former Rocky Flats plutonium facility where from 1952 to 1989 plutonium triggers were produced for the hydrogen bombs in the USA nuclear arsenal. Major anti-nuclear protests were held at this site in the 1980s leading, in part, to its closure. Through decades of operation, much plutonium and other toxic chemicals were released into the local area, which is now a dedicated wild life refuge. We provide some of the history of Rocky Flats and even dig into some of the science behind the building of nuclear bombs. However, given the magnitude of significance, we also aim to provide space for reflection as supported by the audio nuances captured by our field recorder. Duration: 27:15.
[Coordinates: 39.883312, -105.235203]
Online References: NuclearCarePartners.com
RockyFlatsNuclearGuardianship.org
https://www.fws.gov/refuge/rocky_flats/
Show Transcripts: Chem101BPP05a
Chem 101: The Big Picture Podcast, Chapter 05b, Radioactivity
Chapter 05b (Episode 08): Radioactivity
Show Notes
For this “Do the Review” episode we review the first half of the nuclear chapter from your textbook and Conceptual Academy. Topics include the nature of radioactivity, the behavior of the atomic nucleus, transmutation, radioactive half-life, and radioactive dating. Duration: 37:45.
Transcripts (pdf):
Transmutation Worksheet:
Chem 101: The Big Picture Podcast, Chapter 05c, Fission and Fusion
Chapter 05c (Episode 09): Fission and Fusion
Show Notes
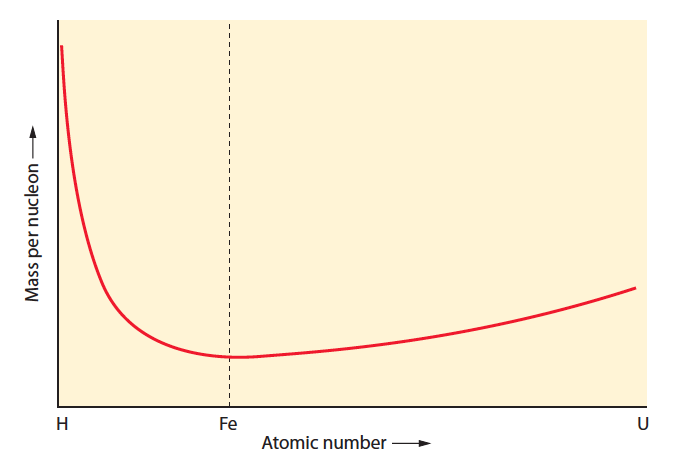
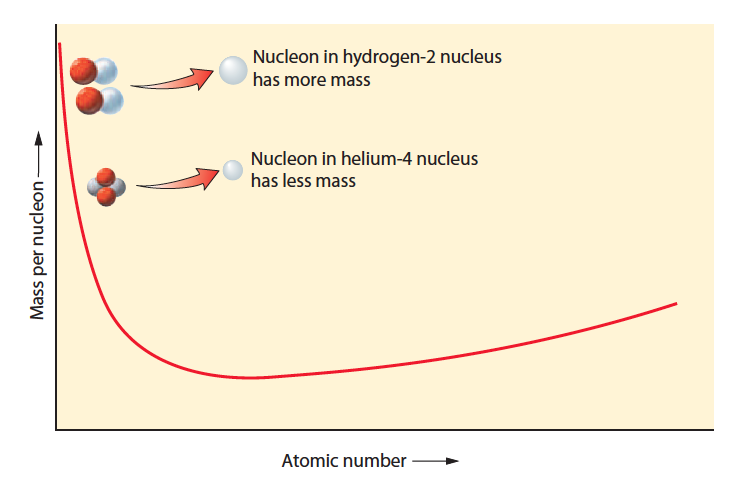
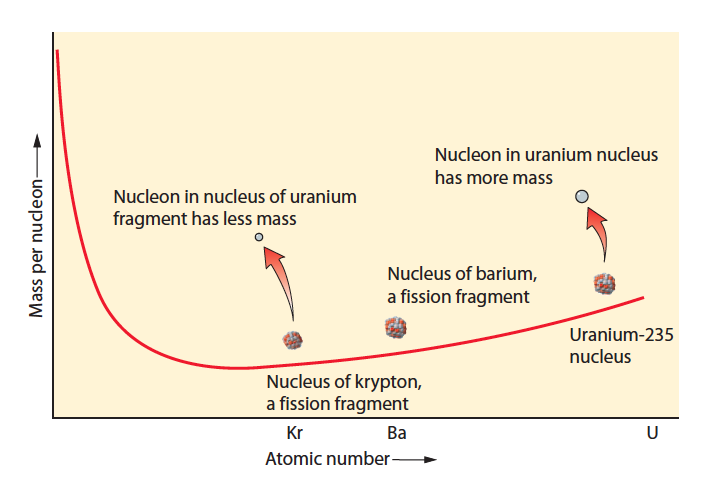
For this “Do the Review” episode we review the second half of the nuclear chapter from your textbook and Conceptual Academy. This includes a focus on the concepts of nuclear fission and fusion. See below for images of the “most important graph in the universe”, which we describe starting at minute 32. Total duration: Duration: 47:50.
Transcripts (pdf):
Most Important Graph in the Universe:
Graph showing the fusion of hydrogen to helium (from left to right, note mass decrease of nucleons):
Graph showing fission of uranium to two lighter daughters (from right to left, note mass decrease of nucleons):
Chem 101: The Big Picture Podcast, Chapter 06A, Rocket Learning
Chapter 06a (Episode 10): Rocket Learning
Show Notes
For this backstory episode, we interview Mike Lucas, rocket engineer, on the nature of his work environment while also collecting his expert study tips for the aspiring college student. Through our conversations, we stumble upon “Step 3 Learning”, which involves synthesizing new information and solutions based upon a foundation of understanding laid down by Step 1 Learning (input) and Step 2 Learning (output) presented in earlier episodes. We explore how the college experience tends to focus on these first two steps. This can be grueling, but it sets the stage for a much more enjoyable career where Step 3 is the primary focus. Duration:30:19.
Transcripts (pdf):
Chem 101: The Big Picture Podcast, Chapter 06B, Ionic Bonding
Chapter 06b (Episode 11): Ionic Bonding
Show Notes
For this “Do the Review” episode, we explore the nature of the ionic bond, which is what forms when an atom that easily loses electrons is placed beside an atom that likes to gain electrons. Topics include the shell model, electron-dot structures, and effective nuclear charge. We also spend a bit of time answering off-topic questions from guest co-host JW. With this foundation, the discussion is extended to include the nature of the metallic bond. Duration: 49:47.
Transcripts:
Chem 101: The Big Picture Podcast, Chapter 6C, Covalent Bonding
Chapter 06c (Episode 12): Covalent Bonding
Show Notes
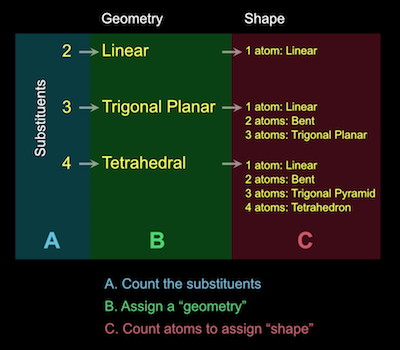
A detailed review of the covalent bond, one-on-one with your textbook author featuring you as our special co-host. Topics include: covalent bond formation, molecules, multiple bonds, VSEPR, substituents, molecular shape, polar bonds, and molecular polarity. The duration is relatively long because the pace is slow but methodical and guaranteed to improve your understanding and appreciation of this foundational chemistry. Duration: 1:08:13.
To the right is an algorithm you can use to determine the shape of a molecule. For example, methane, CH4 has four substituents, which leads to a tetrahedral geometry. Because all four substituents are atoms, the shape of the molecule is a tetrahedron.
Transcripts:
Chem 101: The Big Picture Podcast, Chapter 07A, Online Learning
Chapter 07a (Episode 13): Online Learning
Show notes
Co-hosts Stephanie Blake and John Suchocki interview online learning experts Amy Schneider and Jacque Harris of Ozarks Technical College. Many tips for succeeding in an online course are presented. But we first delve into the many challenges specific to this format. These are challenges that instructors and students should understand before launching into this still developing method of teaching and learning. Duration: 34:16.
Transcripts:
Chem 101: The Big Picture Podcast, Chapter 07B, Molecular Interactions
Chapter 07b (Episode 14): Molecular Interactions
Show Notes
In this “Do-The-Review” episode, co-hosts John and Tracy Suchocki review the four main types of molecular interactions. This includes the dipole-dipole interaction as well as the ion-dipole, dipole-induced dipole, and induced dipole-induced dipole interactions. This is followed by a discussion of the many terms associated with solutions, such as solute, solvent, solubility, concentration, and the mole. The episode concludes with a discussion of reverse osmosis. Duration: 1:01.
Student cartoons of “Deoxygenated Water”
Transcripts:
Chem 101: The Big Picture Podcast, Chapter 08A, The Chemistry of Braces
Chapter 08a (Episode 15): The Chemistry of Braces
Show Notes
Co-hosts John Suchocki, John Wright and Carolyn Wright interview Anoop Sondhi, D.D.S., inventor of the Sondhi Rapid-Set Indirect Bonding System, which has revolutionized the practice of orthodontics. If you’ve worn braces recently, then you’ve likely benefited from Dr. Sondhi’s efforts. We begin with an overview of the chemistry behind his indirect bonding system. This is followed by a discussion of the challenges of developing and then promoting his invention. Duration: 19:47.
Anoop Sondhi, D.D.S.
Transcripts: Podcast 08A
Chem 101: The Big Picture Podcast, Chapter 08B, Water Chemistry, Part 1
Chapter 08b (Episode 16): Water Chemistry, Part 1
Show Notes
Co-hosts John Suchocki and Marjorie Suchocki review the first half of the Conceptual Chemistry chapter on the properties of water. Topics include the open crystalline structure of ice, why ponds freeze from the top down, how salt melts ice, and the properties of liquid water at its surface involving surface tension and capillary action. We review all these topics from the molecular point of view. Duration: 44:09.
Transcripts: Podcast08B
Chem 101: The Big Picture Podcast, Chapter 08C, Water Chemistry, Part 2
Chapter 08c (Episode 17): Water Chemistry, Part 2
Show Notes
Co-hosts John Suchocki and Marjorie Suchocki review the second half of the Conceptual Chemistry chapter on the properties of water. Topics include evaporation, boiling, pressure cookers, hurricanes, specific heat, global warming, and the heat of vaporization. We review all these topics from the molecular point of view. Duration: 42:57.
Transcripts: Podcast08C
Chem 101: The Big Picture Podcast, Chapter 09A, The Value of Communication
Chapter 09A (Episode 18), The Value of Communication
Show Notes
While on the road, host John Suchocki interviews Stephanie Kellar, a senior engineer of the City of Encinitas, California. Stephanie describes how college helped prepare her for a career where she was surprised to find communication skills to be far more critical than technical skills. She describes her experience with the Beacon’s Beach staircase project, proposed as a remedy to a landslide prone path down a sea bluff but met with much resistance from the community. Duration: 25:37.
Photo: Tim Buss
Newspaper Article: The Hartford Courant
Transcripts: Podcast 09A
Chem 101: The Big Picture Podcast, Chapter 09B, Chemical Reactions, Part 1
Chapter 09B (Episode 19), Chemical Reactions, Part 1
TBD
Chem 101: The Big Picture Podcast, Chapter 09C, Chemical Reactions, Part 2
Chapter 09C (Episode 20), Chemical Reactions, Part 2
TBD
Chem 101: The Big Picture Podcast, Chapter 10A, Experiential Learning
Chapter 10A (Episode 21), Experiential Learning
Show Notes
Co-hosts John and Tracy Suchocki interview Peter and Linda Jeschofnig, founders of Hands-on Lab, a company that grew from an environment friendly micro-scale chemistry kit the two of them developed for Peter’s chemistry course back in the early 1990s. Peter and Linda describe their success story of creating a multi-million dollar business that started with creating and assembling lab kits in their garage. Topics include: experiential learning, the value of doing science to learn science, business building tips, and critical thinking skills. Duration: 48:25.
Transcripts: TBD
Chem 101: The Big Picture Podcast, Chapter 10B, Acids and Bases, Part 1
Chapter 10B (Episode 22), Acids and Bases, Part 2
TBD
Chem 101: The Big Picture Podcast, Chapter 10C, Acids and Bases, Part 2
Chapter 10C (Episode 23), Acids and Bases, Part 2
TBD
Chem 101: The Big Picture Podcast, Chapter 11A, Solar Fuels
Chapter 11A (Episode 24), Solar Fuels
Show Notes
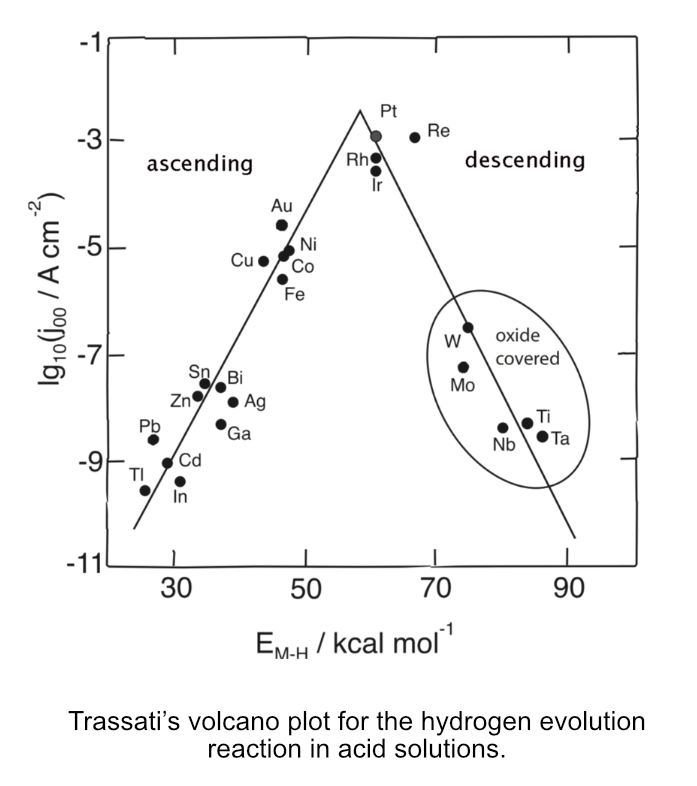
Co-hosts John and Tracy Suchocki interview professor Raj Rajeshwar, one of the leading researchers in the exciting area of solar fuels, which are fuels generated directly from sunlight. We explore the chemistry behind the many challenges still faced in the development of solar fuels. We talk about the prospects. About the possibilities. This is perhaps one of our more technical episodes. It assumes the listener has a basic understanding of chemical reactions, particularly oxidations and reductions. But for all listeners, much value and insight is provided. Duration: 47:39.
Podcasters: Raj, Tracy, and John
The infamous “Volcano Plot” showing platinum and other precious metals within a zone of electrode perfection.
Transcripts: Podcast 11A
Chem 101: The Big Picture Podcast, Chapter 12A, Fossil Fuels
Show Notes
John and Tracy interview Rick, an experienced chemical engineer within the fossil fuel industry who walks us through the process of producing gasoline and other petroleum products from crude oil—the raw material extracted from the ground. Along the way, we learn the state of the industry in a time of diminishing reserves, expanding energy demands, alongside increasing environmental awareness. Duration: 1:02.
A video about our podcasts!